
GLOCARE LABS
Contract Research Organization

Glocare Labs Pvt. Ltd. is a leading Contract Research Organization (CRO), established in 2017 and headquartered in Chennai, India. We specialize in delivering high-quality, end-to-end clinical research services to the pharmaceutical, biotechnology, and healthcare industries.
Committed to scientific excellence, regulatory compliance, and client satisfaction, we partner with our clients throughout every stage of the clinical development.
At Glocare Labs, we blend deep domain expertise with innovative solutions to accelerate the development of safe and effective therapies - ultimately contributing to improved global health outcomes.








Comprehensive clinical research solutions tailored to meet diverse needs with the highest quality standards.

We offer comprehensive clinical research solutions tailored to meet the diverse needs of our clients. Our integrated services span the full clinical studies, ensuring high-quality outcomes and timely project execution aligned with global regulatory standards.
Read Article
Our state-of-the-art Clinical Pharmacology Unit is equipped with 60 beds, designed for maximum flexibility in conducting a wide range of clinical studies ensuring compliance with ethical standards.
Read Article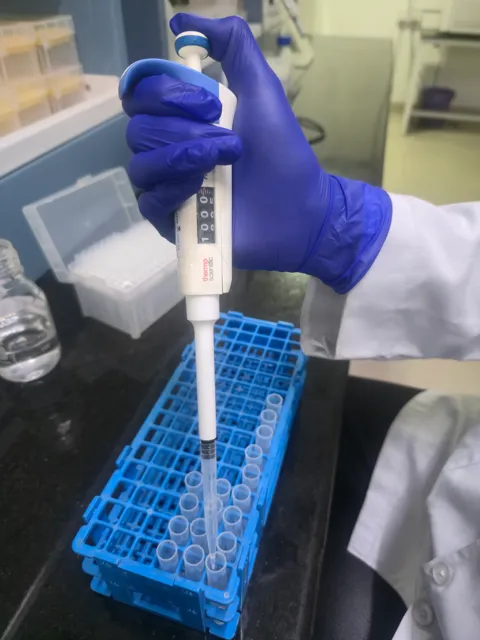
Our Bioanalytical Laboratory is a core component of Glocare Labs clinical research infrastructure, designed to deliver precise, reliable, and regulatory-compliant analysis of drug concentrations in biological matrices.
Read ArticleDiscover our complete range of clinical research services
Our commitment to excellence is validated through international standards and regulatory approvals, ensuring the highest quality in clinical research.
Instituto de Salud Pública de Chile
Our clinical studies have been successfully audited and approved by ISP Chile, affirming our capability to conduct trials in accordance with stringent Latin American and global regulatory standards.
Drugs Controller General of India
Approved by the Drugs Controller General of India (DCGI) for conducting BA/BE studies, ensuring full compliance with Indian regulatory and GCP guidelines.
European Medicines Agency
EMA submission completed – Approval from the European Medicines Agency is currently pending, demonstrating our preparedness and commitment to entering regulated EU markets.
Explore our complete range of international certifications and regulatory approvals
Let's collaborate to advance your clinical research goals. We welcome inquiries about our services and potential partnerships.
No. 2A, 4th Street, Ganga Nagar,
Kodambakkam, Chennai - 600 024, India.
General & Medical:
md@glocarelabscro.comMarketing & Partnership:
marketing@glocarelabscro.comVisit our complete contact page for more ways to reach us