
Bioavailability and Bioequivalence Studies
We offer comprehensive clinical research solutions tailored to meet the diverse needs of our clients. Our integrated services...

Clinical Unit
Our state-of-the-art Clinical Pharmacology Unit is equipped with 60 beds, designed for maximum flexibility in conducting a wide range of clinical studies ensuring ...
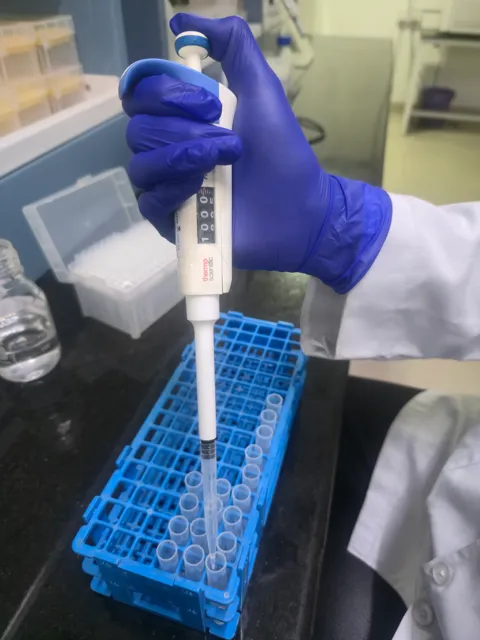
Bioanalytical Laboratory
Our Bioanalytical Laboratory is a core component of Glocare Labs clinical research infrastructure, designed to deliver precise, reliable, and regulatory-compliant analysis...

Clinical Diagnostic Laboratory (CDL)
Our Diagnostic Laboratory at Glocare Labs is NABL accredited National Accreditation Board for Testing and Calibration...

Medical Writing
Our Medical Writing Team plays a critical role in supporting Bioavailability/Bioequivalence (BA/BE) studies with clear, accurate, and regulatory-compliant...

Biowaiver Studies & In Vitro Dissolution Services
At Glocare Labs, we offer specialized biowaiver study services for generic drug development under BCS-based regulatory pathways...

Pharmacovigilance
We provide comprehensive Pharmacovigilance (PV) services to ensure the safety and efficacy of pharmaceutical products throughout their lifecycle...

Regulatory Support Services
We deliver end-to-end Regulatory Support services designed to accelerate approvals, ensure compliance, and streamline...

